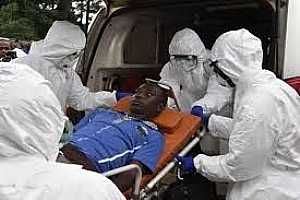
Health of millions at risk from Pakistan floods
Saturday, 21 August 2010
19 August 2010 -- The floods in Pakistan have affected over 15 million people. More than 200 hospitals and clinics have been damaged or destroyed making access to health care difficult. This photo gallery illustrates some of the ways WHO is coordinating the international health response.

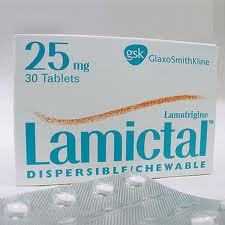
FDA: Aseptic Meningitis Risk with Use of Seizure Drug Lamictal
Saturday, 14 August 2010
The U.S. Food and Drug Administration today warned that the drug Lamictal (lamotrigine), approved to treat seizures and bipolar disorder, can cause aseptic meningitis, an inflammation of the protective membranes (meninges) that cover the brain and spinal cord not caused by bacterial infection.


FDA approves ella™ tablets for prescription emergency contraception
Saturday, 14 August 2010
The U.S. Food and Drug Administration today approved ella™ (ulipristal acetate) tablets for emergency contraception. The prescription-only product prevents pregnancy when taken orally within 120 hours (five days) after a contraceptive failure or unprotected intercourse. It is not intended for routine use as a contraceptive.

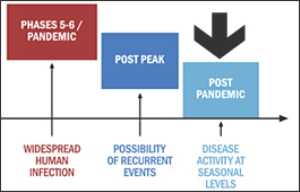
H1N1 in post-pandemic period
Wednesday, 11 August 2010
10 August 2010 -- WHO Director-General, Dr Margaret Chan announced that the H1N1 influenza virus has moved into the post-pandemic period. However, localized outbreaks of various magnitudes are likely to continue.


FDA warns of Salmonella risk from frozen rodents fed to reptiles
Tuesday, 10 August 2010
The U.S. Food and Drug Administration is warning U.S. and international customers who may have purchased frozen mice from Biggers and Callaham LLC, doing business as MiceDirect, that these products, which are used as food for reptiles, have the potential to be contaminated with Salmonella.


July 27, 2010: Former CEO of Seafood Importing Corporation Sentenced to Federal Prison for Importing Falsely Labeled Fish
Saturday, 07 August 2010
NEWARK, N.J. - Thomas George, the former Chief Executive Officer of Sterling Seafood Corporation, was sentenced today to 22 months in prison for importing falsely labeled fish from Vietnam and evading over $60 million in federal tariffs, as well as selling over $500,000 in similarly misbranded fish purchased from another importer, United States Attorney Paul J. Fishman announced.


FDA Seeks Injunction Against Colorado Manufacturer of Cultured Cell Product
Saturday, 07 August 2010
The U.S. Food and Drug Administration is seeking an injunction in federal court against Regenerative Sciences LLC, of Broomfield, Colo., citing violations of current good manufacturing practice (cGMP) that cause its cultured cell product to be adulterated. The product is also misbranded due to the lack of adequate directions for use and the failure to bear the “Rx only” symbol.


All forms of asbestos can cause cancer
Saturday, 07 August 2010
6 August 2010 -- Asbestos has been widely used in buildings and certain commercial products. As a result, 125 million people worldwide are exposed to asbestos at work. Occupational exposure to asbestos causes more than 107 000 deaths annually from lung cancer, mesothelioma and asbestosis.


FDA Issues Assessments of the 510(k) Program and Use of Science in Decision-Making
Thursday, 05 August 2010
The U.S. Food and Drug Administration today issued two comprehensive evaluations containing recommendations that address three key objectives of the agency’s public health mission as it relates to medical devices – foster device innovation, create a more predictable regulatory environment, and enhance device safety.


United States Seeks Permanent Injunction Against New York Food Processor
Tuesday, 03 August 2010
The U.S. Department of Justice, in an action initiated by the U.S. Food and Drug Administration, is seeking a permanent injunction against NY Gourmet Salads, Inc., a processor of ready-to-eat deli salads, seafood salads, and cream cheeses in Brooklyn, N.Y., and Leonard F. Spada, the company's president.
© Copyright 2001-2022 eDoctorOnline.com