FDA Warns Users about Faulty Components in 14 External Defibrillator Models
Wednesday, 28 April 2010
About 280,000 external defibrillators used worldwide in health care facilities, public places, or in the home may malfunction during attempts to rescue people in sudden cardiac arrest, the U.S. Food and Drug Administration warned today.


FDA Changes Process for Medical Device Advisory Committees
Wednesday, 28 April 2010
The U.S. Food and Drug Administration today announced that it will change the way its expert panels review and discuss data and information during public hearings on medical devices under review for premarket approval, effective May 1, 2010.

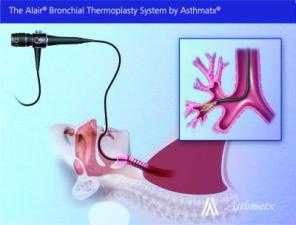
FDA Approves New Device for Adults with Severe and Persistent Asthma
Wednesday, 28 April 2010
The U.S. Food and Drug Administration today approved the first medical device that uses radiofrequency energy to treat severe and persistent asthma in certain adults.


FDA Launches Initiative to Reduce Infusion Pump Risks
Saturday, 24 April 2010
The U.S. Food and Drug Administration today announced a new initiative to address safety problems associated with external infusion pumps, which are devices that deliver fluids, including nutrients and medications, into a patient’s body in a controlled manner.

Immunization week to kick off in 112 countries
Saturday, 24 April 2010
For the first time, immunization campaigns are being launched simultaneously tomorrow in 112 countries and territories in the World Health Organization Regions of the Americas, Eastern Mediterranean and Europe, with the goals of expanding immunization coverage and raising awareness of the importance of vaccines.


Dengue and primary care: a tale of two cities
Friday, 23 April 2010
Dengue virus infection is increasingly being recognized as the world’s major emerging tropical disease. The disease infects an estimated 50 to 100 million people annually. This article of the WHO Bulletin contrasts the neighbouring Brazilian cities of Rio de Janeiro and Niterَi, providing an interesting case study to look at the factors that may be influencing the infestation rate.


FDA Obtains Consent Decree to Stop Marketing of Unapproved Sterilization System
Friday, 23 April 2010
The U.S. Food and Drug Administration today announced that a consent decree has been filed against the device manufacturer, STERIS Corp. of Mentor, Ohio. The decree stops the company from distributing unapproved and misbranded devices used to sterilize heat-sensitive instruments and medical devices. It prohibits the distribution of the STERIS System 1 Processor, or SS1, a tabletop liquid chemical system marketed to disinfect certain devices, such as endoscopes, that cannot withstand steam sterilization.


FDA Issues Statement on IOM Sodium Report
Friday, 23 April 2010
Today’s average sodium intake is several times what the body requires and its long-term effect on our health is very serious. Hypertension, or high blood pressure, affects one in three U.S. adults – nearly 75 million people aged 20 or older. An additional 50 million adults suffer from pre-hypertension. High blood pressure can increase the risk for heart attacks, strokes, heart failure, and kidney failure. Too much sodium in the daily diet is a major contributor to high blood pressure.


FDA to Address Challenges of Using Complex Medical Devices in the Home
Friday, 23 April 2010
The U.S. Food and Drug Administration today announced a new initiative to ensure that caregivers and patients safely use complex medical devices in the home.


FDA Announces Draft Revised Guidance on Transparency and Advisory Committees
Friday, 23 April 2010
The U.S. Food and Drug Administration today announced draft guidance that would expand transparency and disclosure when the agency grants a conflict of interest waiver to permit an individual’s participation at an FDA advisory committee meeting.
© Copyright 2001-2022 eDoctorOnline.com